Ozempic is Brainwashing you (and Not in a Good Way)
GLP-1 agonists suppress the same circuit that drives your ambition, sex drive, and will to build.
Pharmaceutical companies are currently running clinical trials to use semaglutide as a treatment for cocaine addiction, alcohol use disorder, and nicotine dependence. They’re running cocaine trials on your weight loss drug.
At a glance this sounds like a good thing. But think about what it means for a drug to suppress cocaine-seeking behavior in addicts. Do you think one-shotting one of the most dopaminergically hardwired addictions comes without cost?
This means the drug is doing something specific to the dopamine circuitry that drives reward-seeking (and gives you actual reward). That same circuit governs your motivation to train, your drive to build something, your ambition, your sex drive, your ability to feel reward from accomplishment. Its the same system.
I mentioned this on Twitter and was genuinely surprised by how many people took personal offense at it, which leads me to believe that GLPs are also atrophying key brain regions involved in emotional regulation.
In all seriousness, I push back on GLPs not because I think they’re inherently bad nor because I want to be a contrarian. I do so because we are seeing a perfect storm of mass-adoptive hysteria. You must understand that we live in a messianic vacuum. Everybody is struggling, sick, and grasping for something to praise as savior. More often than not that savior comes in the form of health. Diets become cults (think carnivore, peaters, vegans), drugs become religious artifacts, health influencers become priests. Technology, particularly biotech, is occupying the role once reserved for mysticism, theology, and shamanism. You used to be saved by the divine, now you’re saved by a few pills and an insulin syringe. This sets off generational hardwiring that makes you transcend reason (a prerequisite for religiosity).
And honestly I don’t blame them. GLPs are close to magic for the average American (average body fat percentage in the US is 32% btw). We’re hardwired to get what we want with the least amount of effort. Why exercise and eat clean when you can simply slip a tiny little needle into the subcutaneous layer of one of your many fat rolls?
GLP-1s are big-pharma’s wet dream:
Massive TAM (everybody’s fat)
Large rebate margins
Regulatory greenlight
Doesn’t effect root cause (wouldn’t want to cannibalize customer base)
Requires perpetual treatment
Novo Nordisk (creator of Wegovy and Ozempic) became Europe’s largest COMPANY. Not pharmaceutical company. Company in general. The economic winner from the continent that invented modern civilization is a fat loss powder.
Obesity is multi-causal, but really comes down to:
A dysregulated metabolism
A dysregulated reward system
We live in a world that makes us fat and dysfunctional. And thats a huge part of the problem. But we also live in a world that enables unchecked reward seeking behavior. The constant desire for “more” is what drives our economy. This is just gluttony and lust. We want more and we want different. These used to be called sins now they’re sections in college consumer behavior class (not joking).
The instatiable desire for more and better has lead to newer and newer drugs. More mechanisms, more receptor activity, more results. It started with Exenatide, then Liraglutide, then Semaglutide, then Tirzepatide, now Retatutride. Each of these exhibit stronger and stronger fatloss, energy expenditure, and appetite suppression effects.
One day they’ll invent a drug that removes all want entirely. Addictions? old news. Food? waste of time. Self-defined success? not really feeling it. Risk? I simply don’t want enough to go through the effort.
What I’m Actually Saying
Do I think GLP-1s are one-shotting your chance at success and meaning? No. Do I think we need to approach them with nuance? Yes, please. This isn’t some fun experimental research chemical anymore. GLP-1 are one of the fastest drug adoption curves in modern medicine.
If there’s one system you don’t want to mess with, its your dopaminergic system. This is your drive, your will to power, your thumos, your jing essence.
SSRIs one-shotted my dopaminergic system and I still struggle with anhedonia to this day. I’m not saying these drugs are intentionally peddled to make us subservient, docile, and borderline lobotomized, I’m saying those peddling them couldn’t care less if they do (unintended benefit for them).
The pushback I got for saying SSRIs were not worth their squeeze was similar to what I got for saying GLP-1s may have a negative impact on dopaminergic signaling. Same argument but first group had blue hair and pronouns in bio.
I just want to throw this stuff out there to let it marinate. I don’t think GLPs are on the same playing field as SSRIs, and if you have a crippling cocaine addiction or are morbidly obese, the juice is worth the squeeze here. But most people online are neither addicts nor comically fat. They are health-conscious high performers looking for an extra edge. We are blurring the lines between sick-care and performance enhancement. And thus must have different discussions.
People find this particularly hard to believe, so I will spend the rest of the article discussing mechanisms and studies I have built this belief off of.
What Dopamine Actually Does
Before getting into the mechanism, it’s worth being precise about what dopamine actually does. Most people have a vague understanding of it as a “feel good chemical” and that framing undersells the problem.
Dopamine doesn’t make you feel pleasure. What dopamine does is make you want things. It’s the anticipatory signal. It fires before the reward, not during it. It’s what makes you get up in the morning with somewhere to be. It’s what creates the drive to pursue a goal before you’ve achieved it. It’s what makes food look appetizing, sex compelling, money worth working for, and a difficult project feel worth starting.
When dopamine function is suppressed, you don’t necessarily feel bad. You feel flat. Indifferent. Things that used to matter stop mattering. The want goes quiet. You can still function, you just stop being driven by anything.
This is exactly what some GLP-1 users are reporting and exactly what the mechanism predicts.
The Actual Mechanism Behind All This
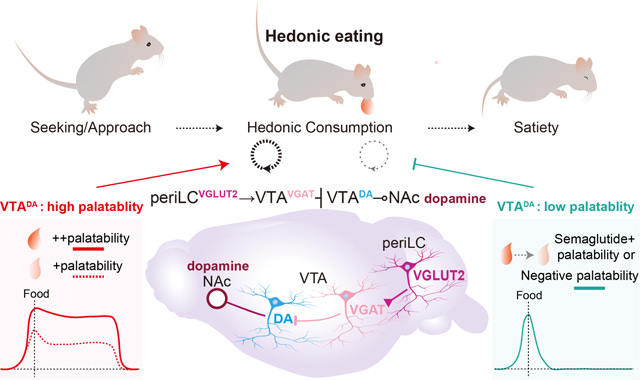
GLP-1 receptors are expressed throughout the brain’s reward system: the ventral tegmental area (VTA), the nucleus accumbens, and the lateral septum. The lateral septum shows the highest GLP-1 receptor density of any brain region tested, higher than anywhere in the gut or periphery. These are not incidental structures. They’re the core of the mesolimbic dopamine pathway: the circuit that produces drive and assigns reward value to experiences.
A 2025 paper published in Science mapped exactly what semaglutide does when it reaches this system. The drug activates GABA neurons in the VTA. GABA neurons are inhibitory: their function is to suppress the firing of other neurons. When semaglutide hits the GABA neurons in the VTA, it increases their inhibitory output onto the dopamine neurons. Dopamine production drops. Less signal reaches the nucleus accumbens. Less reward response. Less drive.
To confirm this was the mechanism and not structural brain change, the researchers restored VTA dopamine activity in mice that had been given semaglutide. Reward-seeking returned immediately. The circuit was intact. The drug had been pharmacologically suppressing it. Remove the suppression, the drive comes back.
A second 2025 paper in Science Advances confirmed the circuit in the context of cocaine. GLP-1R activation increased VTA GABA neuron activity and decreased VTA dopamine neuron activity in rats trained to seek cocaine. The researchers mapped the specific anatomical projection: GLP-1-producing neurons in the nucleus tractus solitarius (NTS) send direct projections to the VTA, where they activate those inhibitory GABA neurons. This is a documented anatomical circuit with a real function in your brain. The drug is co-opting it.
The Suppression Is Not Food-Specific
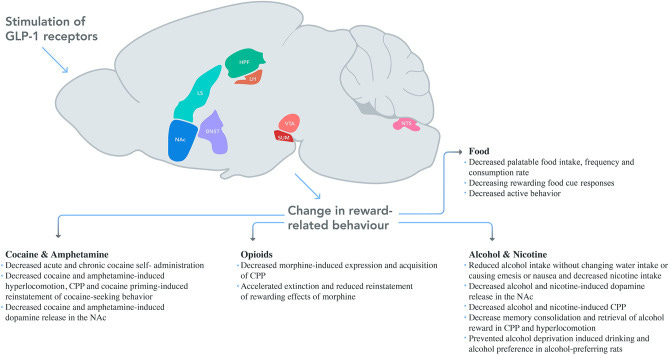
A systematic review of 101 studies (30 clinical, 71 preclinical) found GLP-1 agonists reduce reward-seeking behavior across palatable food, cocaine, amphetamine, alcohol, and nicotine through the same shared mechanism. One pathway, every reward category tested. The drug doesn’t distinguish between an unhealthy craving and a healthy motivational drive. Both run through the same dopamine circuitry.
The human data confirms this extends to non-substance rewards. A 2021 randomized controlled trial gave liraglutide to volunteers and measured incentive motivation for food and for monetary rewards. Under normal conditions, hunger amplifies motivation for both: biological drive state increases how hard you’ll work for any reward, not just food. That’s a well-established effect across decades of research.
After liraglutide, that effect was statistically eliminated. Hunger no longer amplified motivation for money. Food and monetary rewards responded identically to the drug. The researchers weren’t measuring appetite suppression. They were measuring motivational drive, and it was blunted across both reward categories equally.
The drug is not suppressing appetite as much as it’s suppressing the motivational amplification system that appetite happens to run through.
The Wanting Problem
To undesrtand whats going on, you must distinguish “wanting” from “liking.” Wanting is dopaminergic: it’s incentive salience, the pull toward a reward before you have it. Liking is opioidergic and serotonergic: the pleasure of the experience itself. They’re separate systems, and they can be affected independently.
What GLP-1 agonists suppress is primarily the wanting signal. The incentive salience that makes a goal feel worth pursuing. Users on these drugs often report that when they do engage with something, they can still enjoy it. The liking is intact. But the wanting that would have pulled them toward it in the first place is diminished. They don’t seek. They don’t initiate. They wait to be brought to the reward rather than going after it.
This is why users describe being “productive but feeling nothing about it.” They can execute. They’ve just lost the internal pull that generates ambition, anticipation, and drive. That distinction between wanting and liking is precisely why the effect is so easy to dismiss. Users don’t feel acutely depressed, they just stop caring. And “I stopped caring” is a much harder symptom to attribute to a medication than “I feel terrible.”
If you still experience a reward response, but have no incentive salience. You are less self-initiating and more externally driven. You are easier to steer with defaults and cues. We live in a world of constant cue bombardment. Remember Operation bluebird? Project Artichoke? It is no longer a conspiracy that our music, movies, and social media feeds are designed to degrade our agency and impact our behavior. Chew on this for me.
The Drift
A population with chronically suppressed dopaminergic drive doesn’t become obviously broken. It becomes gradually less self-directed. Slightly less inclined to hunt for information rather than consume what’s pushed. Slightly less likely to build an opinion from the ground up versus defaulting to whatever has the most social proof. Slightly more comfortable inside existing structure rather than questioning it.
None of this requires conspiracy. Dopamine drives reward prediction error: the signal that says “that was worth it, do it again.” Suppress that signal and the effort-reward cycle weakens. People can still perform tasks. They just stop building the internal momentum that converts effort into drive. Compliance without ignition.
Lower incentive salience inside a world optimized to capture passive attention is just math. The algorithms, the narratives, the institutional defaults, they all flow downhill into low-resistance (low agency) people. Now amplified by drugs.
Of course some people on GLP-1s report more agency after starting, because the food noise was the thing consuming their bandwidth. For that population, suppressing a compulsive loop freed up genuine directed drive. The actual question is whether the drug reduces compulsive wanting while preserving purposeful wanting. For some people, yes. For others, no. The drug doesn’t know which you are. Your doctor isn’t tracking it. In a world saturated with low-effort, high-stimulation options engineered to capture passive attention, I’d rather err on the side of keeping the wanting. Besides, vices like compulsive behavior, insatiable cravings, and risk-taking are also an inherent edge. Your objective should be to harness these impulses, not eradicate them. Lock in.
So you need to ask yourself: Are you really mentally and metabolically compromised enough to roll the dice? Thats your decision to make.
The Prefrontal Cortex Layer
The limbic reward circuit isn’t the only area involved. GLP-1 receptors are also expressed in the prefrontal cortex (PFC), which governs executive function, goal-directed behavior, and the capacity to pursue long-term objectives over short-term comfort.
Beyond dopamine, GLP-1 modulates glutamatergic neurotransmission throughout the brain. Glutamate is the primary excitatory neurotransmitter, and it drives the sustained firing patterns that underlie focused effort, goal maintenance, and the cognitive architecture of motivation. When GLP-1 agonists dampen glutamatergic activity in prefrontal circuits, the effect isn’t just reduced wanting. It’s reduced capacity for the kind of directed, sustained drive that ambitious work requires.
The practical implication is that what users experience as “brain fog” or “reduced ambition” may not be purely limbic in origin. The same drug touching the VTA dopamine circuit is simultaneously modulating the prefrontal executive layer. Both contribute to the experience of motivated behavior, and both are being suppressed. Once again contributing to subtle “unraveling” of your coordinative agency.
Tirzepatide and Retatrutide Make This Worse
Most of the mechanistic research above is on semaglutide and liraglutide. The newer compounds escalate the problem.
Tirzepatide (Mounjaro, Zepbound) is a dual agonist: GLP-1 receptors and GIP receptors simultaneously. GIP receptors are expressed in the VTA and nucleus accumbens and modulate dopamine neurotransmission through pathways that overlap with, but are distinct from, the GLP-1R mechanism. A dual agonist hits the dopamine circuit from 2 angles at once.
Retatrutide, currently in Phase 3 trials at Eli Lilly, adds a third: glucagon receptors. GLP-1, GIP, and glucagon receptors all have CNS expression. All 3 have been documented in reward-relevant brain regions. A triple agonist pressing on 3 receptor populations in the mesolimbic system simultaneously has no pharmacological precedent in humans. The long-term neurological implications haven’t been studied but unless you’re literally Honey Boo Boo’s mom or a bodybuilder with unresolved childhood trauma, this triple agonism is entirely unnecessary
Tirzepatide outperforms semaglutide on weight loss in head-to-head trials. Retatrutide outperforms tirzepatide in early comparisons. The magnitude of reward suppression likely tracks with efficacy. Nobody is measuring it because nobody really cares.
The Disclosure Gap
GLP-1 clinical trials are designed to measure weight loss, glycemic control, and cardiovascular outcomes. Those are the primary and secondary endpoints that determine FDA approval. Motivated behavior, hedonic capacity, libido, and dopaminergic drive are not standard endpoints in these trials. They’re not measured. If an effect isn’t measured, it doesn’t appear in the label. This is allopathic model 101.
This is structurally different from a side effect being studied and found to be acceptable. The effect isn’t studied. The trials are not set up to detect it. The mechanism research exists in separate academic literature on reward neuroscience and addiction pharmacology, authored by researchers who are largely not in conversation with the clinicians prescribing these drugs for weight loss.
The result is that a drug with a well-characterized mechanism of dopamine suppression carries a label that says nothing about dopamine, motivation, or reward, because the approval data was never designed to capture those outcomes.
Modern medicine continues to operate trials with mechanistic myopia because that is what the entire model is predicated on. This will not change. We think we know everything there is to know about biology. Always have and always will (remember when they put asbestos in cigarette filters?). Maybe its hubris, maybe its indifference. Probably both.
What People Are Actually Experiencing
The anecdotal reports have been building for two years and follow a pattern consistent enough to be clinically interesting.
Users describe the food cravings going quiet, which is what gets marketed. What gets less coverage is what else goes quiet alongside it. Reports of alcohol use dropping dramatically are common enough that addiction researchers launched the cocaine and alcohol trials. Reports of impulsive spending disappearing, gambling urges evaporating, and nicotine cravings stopping without trying are widespread.
All good things right?
The benefit of dropping lifelong bad behaviors outshines the subtle changes in personality and quality of life. Hobbies feel flat. Work feels mechanical. Sex drive drops. Ambitions now feel distant. “Nothing feels exciting,” “Things don’t hit the way I used to,”.
These complaints are common enough to have generated sustained discussion among users who got the body they wanted and are now trying to figure out why it doesn’t feel like they thought it would.
The drug suppresses the motivational drive circuit. That circuit doesn’t know the difference between a pathological craving and a goal you’ve spent five years working toward. When you suppress one, you suppress both. The dose is not selective.
The Counterargument (There Always is One)
The counterargument worth taking seriously is that GLP-1 agonists may “normalize” dysregulated dopamine in insulin-resistant patients with severe metabolic dysfunction. Chronic metabolic disease compromises reward circuitry through inflammation and impaired signaling, and there’s legitimate data showing GLP-1 agonists can restore motivational function in that specific population. In a severely compromised system, the drug may bring dopamine function closer to baseline rather than below it.
This is totally true, but that argument applies to a specific clinical population. It doesn’t apply to the majority of current users, who are people without severe insulin resistance trying to lose 15-30 lbs. Their dopamine systems were functioning before they started. The normalization rationale wasn’t built for them, and it’s being used to dismiss a concern that the mechanistic research takes seriously. Sick care and optimization are two different things and must be treated as such.
We’ve (I’ve) Seen this Pattern Before
My SSRI experience produced a functionally similar outcome through a different mechanism. Serotonin reuptake inhibition rather than GABAergic dopamine suppression, but the result overlapped: flat affect, reduced reward response, (not so) subtle degradation of quality of life. No acute distress. Just a persistent absence of the intensity that makes things feel worth doing.
It took me ages to understand what had happened, and longer to fix it. Nobody discussed it before the prescription. The mechanism was documented in the literature it just didn’t make it into the clinical conversation.
The GLP-1 situation is the same script. The weight loss data is disclosed because it sells the drug. The dopamine suppression mechanism is not disclosed because it complicates the sale. The studies exist, the addiction trials are actively running, and the information is in the same journals.
“Low motivation.” “Nothing feels exciting anymore.” “I stopped caring about things I used to care about.” These are not personality changes. They are not life circumstances. They are not depression in the conventional sense. They are the documented, mechanistic downstream effect of pharmacological suppression of dopaminergic drive in the VTA, produced by the same mechanism that pharmaceutical companies are simultaneously running trials to patent as an addiction cure. Not everybody will experience negative effects. For some people, the outcome is worth the risk. But given our track record with SSRIs, Benzos and Opoids, we should probably learn to exercise some scrutiny once and a while.
If those symptoms appeared 4-6 weeks after starting a GLP-1 agonist, you know the mechanism. Your doctor probably won’t bring it up. The good news is research indicates that this dopaminergic countersignaling goes away once you get off the juice. The more you know.
That is all. Godspeed.
Sources for the curious and curious-adjacent
Zhu Z et al. (2025). Hedonic eating is controlled by dopamine neurons that oppose GLP-1R satiety. Science. PMC12009138.
Merkel R et al. (2025). An endogenous GLP-1 circuit engages VTA GABA neurons to regulate mesolimbic dopamine neurons and attenuate cocaine seeking. Science Advances. PMC11864183.
Eren-Yazicioglu CY et al. (2021). Can GLP-1 Be a Target for Reward System Related Disorders? Frontiers in Behavioral Neuroscience. PMC7848227.
Hanssen R et al. (2021). GLP-1 and hunger modulate incentive motivation depending on insulin sensitivity in humans. Molecular Metabolism. PMC7859312.







GLP dick riders might be worse than carnivore types imo
hii- I’m new to substack & saw you were also on the top 100 in health. I’m 29 and have beaten cancer twice. I’m sharing all the tips & tricks I’ve learned on healing my body from the inside out.
Berries are 1 of the top 5 cancer fighting foods, check out my last post!
https://livlifee.substack.com/p/why-i-eat-blueberries-every-day-and?utm_campaign=post-expanded-share&utm_medium=post%20viewer